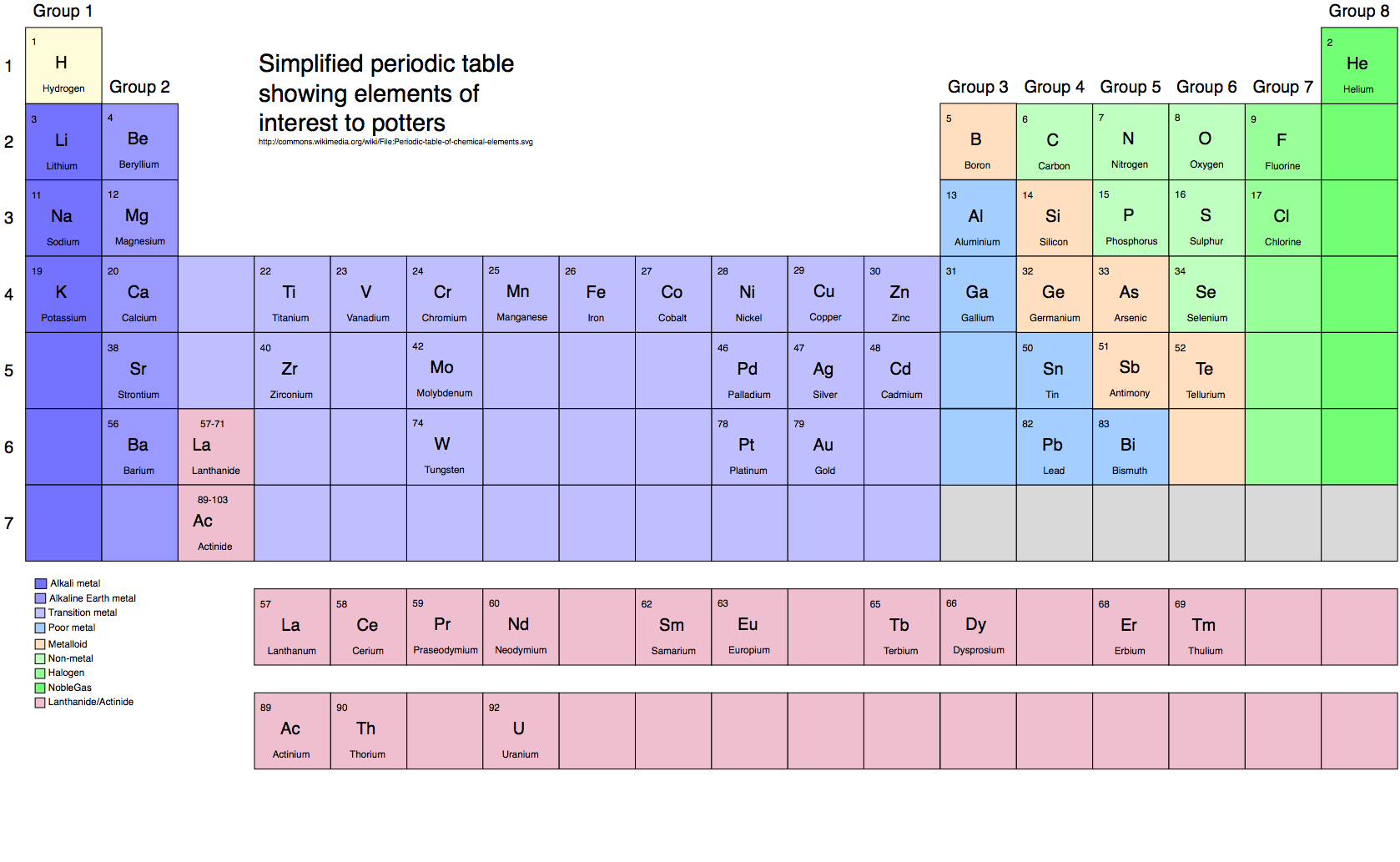
I have been asked to teach several glaze courses in 2017.
A new pottery school is opening in Forest Row in Sussex. I will be teaching glazes there 0n 17-19 February 2017. Contact Katrina Pechal on 0789 444 7938. You can visit the school when they open in December, courses start in January with tutors including Stephen Parry and Ruthanne Tudball.
The founder Katrina Pechal says, “ I am saddened to see so many high-level ceramics courses around the country closing. My days training at Camberwell School of Art were inspiring, having lessons with famous potters like Takashi Yasadu, Colin Pearson and Ewen Henderson among others was a great privilege and I want the next generation of potters to have the same opportunities. I have noticed a recent increase in the popularity of pottery, thanks in part to the BBC’s “The Great Pottery Throw Down. I feel this is a perfect time to move to a bigger site with better facilities where I can help students to train and develop their craft”.
Forest Row School of Ceramics will be based in the Rachel Carson building at Emerson College, set in beautiful grounds within walking distance of the village. It offers outdoor space, which Katrina needs for a kiln site (a kiln-building workshop run by Joe Finch, Potter and author of Kiln Construction (a brick by brick approach), is already planned for early next year). Emerson also has more classrooms Katrina can utilise as she expands plus affordable student accommodation it needed.
Forest Row is three miles from East Grinstead in Sussex, situated next to the idyllic Ashdown Forest yet within easy access of the M23/M25 and a short drive from Gatwick Airport, the South Downs and the creative hub of Brighton.

I will also be teaching colour in glazes at West Dean College on 5-9 March (the course is now full but you can try the waiting list), and demonstrating throwing and glazes all weekend at the Scottish Potters’ Spring Workshop at Tulliallan on 10-12 March.
For anyone in London there are glaze courses at the Morley College in February and April but I will not be teaching them.
Exciting news: I will be teaching a glaze course in Belgium in 2018.
If you can’t come on a course, you can still learn from my books.